Successful drug development requires a deep understanding of the disease of interest—its etiology, epidemiology, presentation, manifestations, and progression. In rare diseases, however, much of this information may be unknown. Patient populations are small and historical data are collected inconsistently and dispersed across treating physicians practicing in diverse geographies. Patient registries and natural history studies are valuable sources of rare disease information for sponsors seeking to design reliable clinical trials with relevant, meaningful outcome measures.
In this blog post, we explore the distinctions between registries and natural history studies, highlighting the potential value of each in informing and shaping clinical development in rare diseases.
Challenges of rare disease development
In 2018, Larissa Lapteva, M.D., MHS, and colleagues outlined the building blocks of every product development program, emphasizing their applicability in rare diseases (see Figure 1).1 The foundation of this pyramid is knowledge of the disease and its natural history.
Figure 1. Building blocks of a product development program1
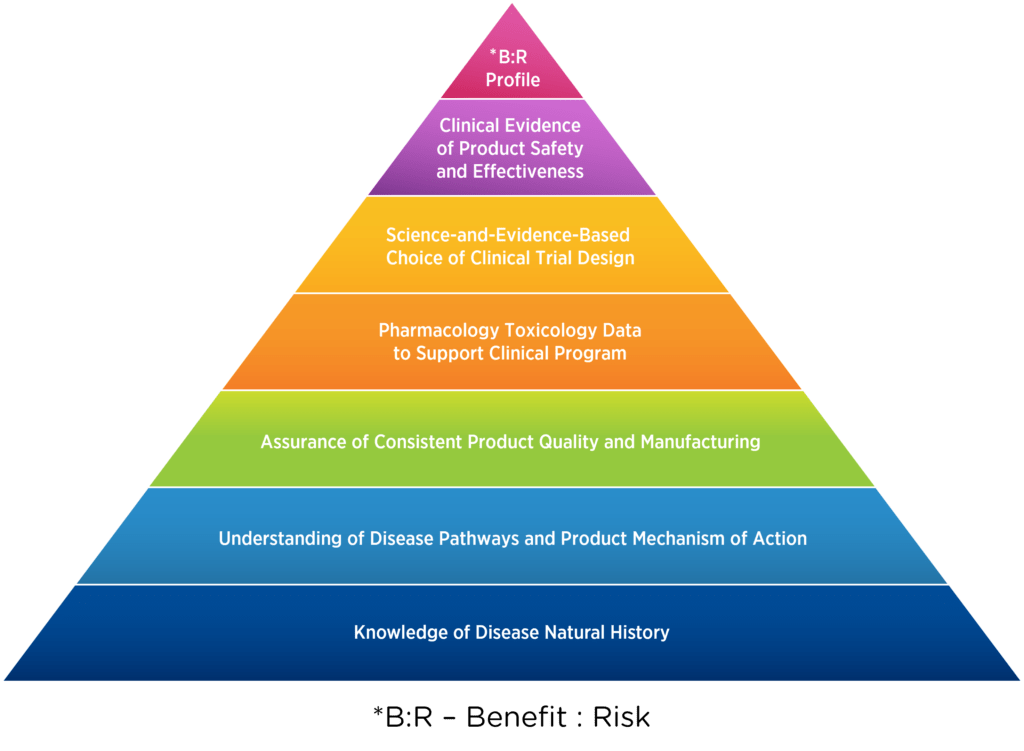
In rare diseases, developing a comprehensive understanding of the disease of interest is hampered by:
- Inherently small populations
- Scarce, incomplete, or inconsistent data
- Disease heterogeneity, which complicates diagnosis, categorization, and data collection
- Lack of precedents
- Need for more careful, more extensive planning
Observational studies play a critical role in addressing these challenges and filling in knowledge gaps.
Overview of observational studies
Unlike clinical trials, where patients receive defined interventions according to a protocol, observational studies do not assign participants to specific interventions and do not attempt to affect the outcome.
Observational studies are divided into two categories:
- Registry studies, which may include a broad collection of defined data
- Natural history studies, which are used for controlled, detailed collection of data that may be subject to regulatory scrutiny
The terms registry study and natural history study are often used interchangeably, but they are not synonymous in either definition or application.
Defining and utilizing patient registries
A patient registry is an organized system for collecting, storing, retrieving, analyzing, and disseminating information on individuals who have one of the following:
- A disease of interest
- A condition or risk factor that predisposes them to a health-related event
- Prior exposure to substances that are known or suspected to cause adverse health effects
Some patient registries are purpose-driven, designed for collection of defined demographic, epidemiological, efficacy, cost-effectiveness, quality of life, or care pattern data. Most registries, however, are less restrictive and less structured and can be set up to collect any data, including patient communications and post-marketing data.
Given that registries are typically broad in scope, registry studies may be useful throughout the drug development pathway. Common applications of patient registries include:
- Collection of disease information
- Advancement of research hypotheses
- Clinical trial recruitment
- Observation or identification of population behavior patterns
- Monitoring of outcomes
- Study of standard of care or best practices
Defining and utilizing natural history studies
Natural history studies document the natural course of a disease, starting just before its inception and progressing through its pre-symptomatic phase and different clinical stages until the patient is cured, chronically disabled, or dead.
Unlike registries, natural history studies are designed with a specific intent, for instance, tracking the course of a disease over time, identifying variables that correlate with the disease and outcomes in the absence of treatment, or informing clinical trial design. These studies may also be used for:
- Obtaining more accurate estimates of prevalence
- Identifying and differentiating among disease subtypes
- Identifying and evaluating potential biomarkers
- Evaluating potential clinical outcome assessments
- Informing protocol design, including study duration, inclusion and exclusion criteria, and appropriate endpoints
Natural history studies can be extremely valuable in rare diseases where the inclusion of a placebo control clinical trial arm may not be feasible. In certain circumstances, a natural history study can serve as a surrogate for the control population, provided the study is designed to meet the requirement for inclusion in regulatory submission.
Types of natural history studies
There are several natural history study designs, each with advantages and disadvantages. The designs may be retrospective, focused on the present, or prospective.
Medical literature reviews are the easiest, least expensive way to begin clarifying the natural history of a disease, but data may be difficult to standardize, and these studies may not meet natural history study objectives. Retrospective chart reviews are also relatively inexpensive, though missing and non-standardized data may be an issue.
Prospective designs include cross-sectional studies and prospective longitudinal studies (see Figure 2). Cross-sectional studies collect data from a variety of patients at a single point in time. While these studies may provide insight into disease generalities, they do not provide information on progression or patient experience. On the other hand, longitudinal studies collect data over a prospectively defined period. These studies can be lengthy and costly, but they allow researchers to assess disease progression over time.
Figure 2. Comparison of prospective natural history study designs

1 Lapteva L, Vatsan R, Purohit-Sheth T. Regenerative medicine therapies for rare diseases. Trans Sci Rare Dis. 2018;3(3-4):121-132.
