Last Updated: January 7, 2026, 9 am UTC
Cancer is the leading cause of death worldwide, accounting for nearly one in every six deaths. According to a recent paper, an estimated 15% of cancer-related deaths could be avoided by early disease detection.1 However, there are currently very few population-based screening programs and, even where a screening test does exist, compliance is relatively low. Liquid biopsy tests—which use non-invasive sampling methods to derive information that can be used to detect cancer—represent both a promising alternative to existing screening methods and an opportunity to develop cancer detection tests for indications where no such screening method is yet available.
In this blog post, we discuss the importance of early cancer detection and explore the current landscape and key considerations for developing liquid biopsies for cancer screening.
The void in early cancer detection
Being able to identify aggressive tumors at an early stage would enable more effective treatment, improving survival and quality of life (see Figure 1).
Figure 1. Colon cancer survival by stage2
At later stages of disease, surgery is less effective, and chemotherapy is often more toxic. The expenses associated with medication, clinical and home visits, and in-hospital care also increase (see Figure 2).
Figure 2. Cancer costs by stage3
Currently, the US Preventative Services Task Force (USPSTF) only recommends population-based screening for colorectal cancer (CRC) and breast cancer in an average-risk population and for lung cancer in a high-risk population. Prostate cancer screening is recommended on an individual basis. Compliance rates for screening vary, but in the US, screening compliance is approximately 70% for CRC and 75% for breast cancer. According to Global Cancer Statistics estimates, more than 60% of cancer-related deaths are caused by malignancies for which there is no screening test available.4
Liquid biopsy has come a long way since its inception
Driven by advances in technology, the global liquid biopsy market is expected to grow at a compounded annual growth rate (CAGR) of nearly 20% over the next 10 years.5 The potential applications of liquid biopsy vary widely, but the four most common are:
- Tumor typing and genetic profiling, where the identification of specific genetic changes is used to inform treatment
- Minimal residual disease (MRD) monitoring, which is used to detect cancer recurrence
- Triage testing, where the liquid biopsy is used for cancer detection or staging in patients who present with symptoms or abnormalities
- Screening for early cancer detection, in which asymptomatic individuals are tested with the objective of reducing overall cancer incidence in the population
Using liquid biopsy for screening and early cancer detection has several potential advantages:
- Minimally invasive compared to tissue biopsy or other screening modalities
- Lower cost and less burden to the healthcare infrastructure
- Higher projected compliance
- Multi-cancer screening from a single sample
- Ability to capture non-tumor derived data—including the immune response—which dominate in the early stages of disease when tumor-related biomarkers in the bloodstream are very low
Liquid biopsies can be carried out quickly and can provide genomic, proteomic, and metabolomic information, enabling a pan-omics approach to early cancer detection. Further, machine learning, a subset of artificial intelligence (AI), can apply mathematical and statistical methods to this information to improve a computer’s performance in decision-making. Using large amounts of data, algorithms can be trained to derive test results, either positive or negative, improving performance in sensitivity and specificity.6
Developing a liquid biopsy test for asymptomatic individuals: What you should know
Liquid biopsy performance revolves around sensitivity, the ability to detect cancer, and specificity, the ability to detect healthy individuals. For cancer screening and early detection tests, sensitivity is critical. Tests with low sensitivity are associated with a high incidence of false negative results, which pose a significant risk to patients. Tests with low specificity are associated with a high incidence of false positive results, which could lead to patients being subjected to invasive tests, overtreatment, and anxiety. It is, however, important to keep in mind that improving the specificity of a test usually results in lowering sensitivity and increasing the incidence of false negatives. Thus, determining an acceptable trade-off between specificity and sensitivity is important for any screening or early detection test.
Challenges of using liquid biopsy in early-stage disease
Many of the existing liquid biopsies focused on early cancer detection lack the sensitivity required for reliable detection of early-stage cancers.7 There are numerous challenges to early cancer detection with in vitro diagnostics:
- Many cancers are asymptomatic in the early stages.
- Tumor-derived genetic biomarkers may not be shed into the bloodstream in the early stages of disease and, even when they are, they may only be present at very low concentrations.8,9
- Certain cancer protein biomarkers—including prostate specific antigen (PSA) and carcinoma-antigen-125 (CA-125)—may not be elevated in patients with cancer, even in advanced disease.10 These biomarkers also lack specificity, as they may also be elevated in patients without cancer.11
Given these challenges, in vitro diagnostics may need to combine tumor derived signals with non-tumor derived information, such as immune response, to be more effective in detecting early-stage cancers.12
Before selecting your liquid biopsy indication, consider these variables
Currently, the most researched biomarkers for cancer detection have been in colorectal and breast cancer, both of which have existing screening paradigms. When selecting an indication for early cancer detection using liquid biopsy, consider the following factors.
Available biomarkers – Do validated biomarkers already exist? Are the biomarkers specific to the indication or are they also present in other conditions or in healthy individuals?
Disease incidence – Incidence affects not only the number of people who would need to be enrolled in a clinical validation study to reach statistical significance, but also the market potential of the test.
Cancer histology – Can the test detect different histology? If not, how aggressive is the screening and treatment plan for that particular cancer?
Screening and treatment paradigms – Does a screening paradigm already exist? If so, there may already be guidance on the sensitivity and specificity required for a screening test. For example, for CRC, FDA guidance recommends 73% sensitivity and 90% specificity. If not, developers would need to identify and establish appropriate thresholds for performance.
Single vs multi-cancer screening claims – Screening for individual cancers can be expensive and, in the future, it may be more cost- and time-efficient to use multi-cancer tests that can detect a range of cancers from a single sample. Such tests would be valuable for triaging patients who present with non-specific symptoms, where the suspicion of cancer is low. However, there are challenges with pursuing a multi-cancer screening claim—for instance, discriminating between tissues of origin or confirming false positives and false negatives. Clinical validation is difficult in an average-risk intended use population, and clinical utility would require extremely high specificity to be practical and cost-effective. Thus, one strategy might be to pursue a single indication, and then add new indications as the test is further developed and real-world evidence is generated.
Developers also need to consider preanalytical variables that may impact the quality of liquid biopsy tests (see Figure 3). Standardizing these variables—and the procedures associated with them—is critical for ensuring that the test is robust and reproducible. Most of this standardization is done with nonclinical studies to evaluate analytical performance, including precision, reproducibility, and stability studies.
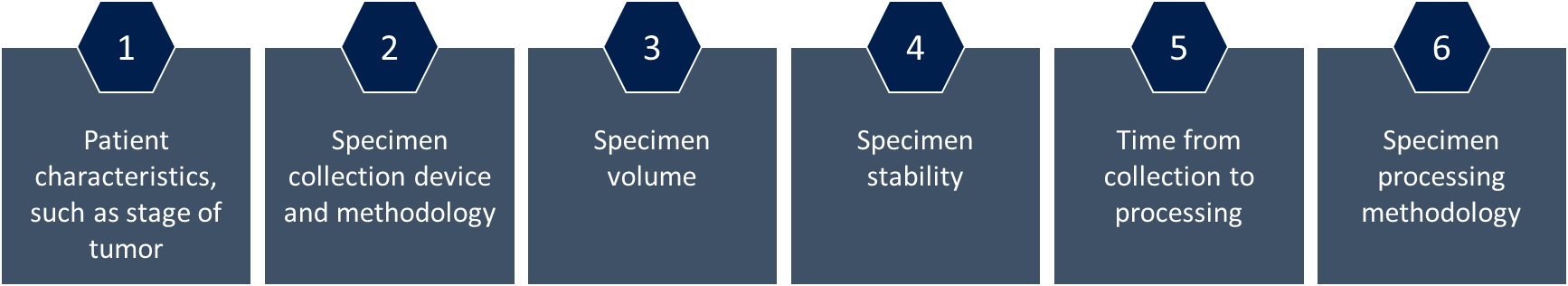
Figure 3. Preanalytical variables that need to be standardized for liquid biopsies13
The future of liquid biopsy is now
Liquid biopsies that can detect early-stage cancers have the potential to improve patient prognosis and survival, but most current techniques still lack the necessary detection capability. Technology continues to evolve, bringing the promise of using a single small sample to detect precancerous lesions or early tumors ever closer.
To learn more about liquid biopsy technology for early cancer detection, watch our on-demand webinar.
[1] Clarke CA, et al. Projected reductions in absolute cancer-related deaths from diagnosing cancers before metastasis, 2006-2015. Cancer Epidemiol Biomark Prev. 2020;29:895–902.
[2] SEER Incidence Data, November 2022 Submission (1975-2000).
[3] Connal S, et al. Liquid biopsies: the future of cancer early detection. J Transl Med 2023;21(1):118.
[4] Sung H, et al. Global Cancer Statistics 2020: GLOBOCAN Estimates of Incidence and Mortality Worldwide for 36 Cancers in 185 Countries. CA Cancer J Clin. 2021;71(3):209-249.
[5] BIS Research. Liquid Biopsy Market – A Global and Regional Analysis. Available at https://bisresearch.com/industry-report/liquid-biopsy-market-report.html.
[6] Huang S, et al. Artificial intelligence in cancer diagnosis and prognosis: Opportunities and challenges. Cancer Lett. 2020;471:61–71.
[7] Klein EA, et al. Clinical validation of a targeted methylation-based multi-cancer early detection test using an independent validation set. Ann Oncol. 2021;32(9):1167–1177.
[8] Wu TM, et al. Power and promise of next-generation sequencing in liquid biopsies and cancer control. Cancer Control. 2020;27(3):107327482093480.
[9] Campos-Carrillo A, et al. Circulating tumor DNA as an early cancer detection tool. Pharmacol Ther. 2020;207:107458.
[10] Bettegowda C, et al. Detection of circulating tumor DNA in early- and late-stage human malignancies. Sci Transl Med. 2014. https://doi.org/10.1126/scitranslmed.3007094.
[11] Dochez V, et al. Biomarkers and algorithms for diagnosis of ovarian cancer: CA125, HE4, RMI and ROMA, a review. J Ovarian Res. 2019;12(1):28.
[12] Crosby D, et al. Early detection of cancer. Science. 2022;375(6586):9040.
[13] Tsui DWY, et al. Development, validation, and regulatory considerations for a liquid biopsy test. Clin Chem. 2020;66(3):408-414.
 Webinar
Webinar 


 Perspectives Blog
Perspectives Blog 