Last Updated: October 9, 2024, 11 am UTC
Rare cancers account for 25-30 percent of all new cancer diagnoses and 25 percent of cancer deaths.1 With their poorly understood natural histories, phenotypic heterogeneity, and diverse clinical manifestations, rare cancers pose challenges to drug development and represent a significant unmet need in oncology. Faced with limited treatment options, researchers, clinicians, and patients may be seeking approaches to accelerate the development and approval of novel therapies. In this blog post, we review the regulatory programs available to expedite treatments for rare disorders and serious diseases.
Accelerating therapies for serious conditions: Where it began
The FDA first formally articulated its thinking on expediting availability of promising new therapies in Title 21 of the Code of Federal Regulations, Section 312, Subpart E. Subpart E regulations are intended to accelerate the availability of new therapies to patients with serious conditions, especially in cases where there are no satisfactory alternative therapies, while still preserving appropriate standards for safety and efficacy.2 The regulations also reflect the agency’s recognition that patients and physicians might be willing to accept greater treatment-related risks for life-threatening or severely debilitating diseases. In addition to providing guidance for early consultation with FDA for efficient trial design, Subpart E references the potential to rely on well-controlled Phase 2 studies for evidence of effectiveness.
The FDA has a history of applying the philosophy behind Subpart E to drugs for rare diseases through its expedited programs. Recognizing that certain aspects of traditional drug development may not be feasible for rare diseases the agency has demonstrated a willingness to be flexible in evaluating drugs to treat these conditions.
How to know if your drug will qualify
The approvals of tazmetostat for advanced epithelioid sarcoma and selumetinib for neurofibromatosis type 1 demonstrate FDA’s commitment to making the unique needs of rare cancers a top priority. For therapies like these, there are several regulatory mechanisms available to help expedite rare oncology programs, including designations (see Figure 1) and an accelerated approval pathway.
Designations

Figure 1. Key features of designations to accelerate development
Eligibility for each of these designations is associated with specific qualification criteria (see Figure 2).
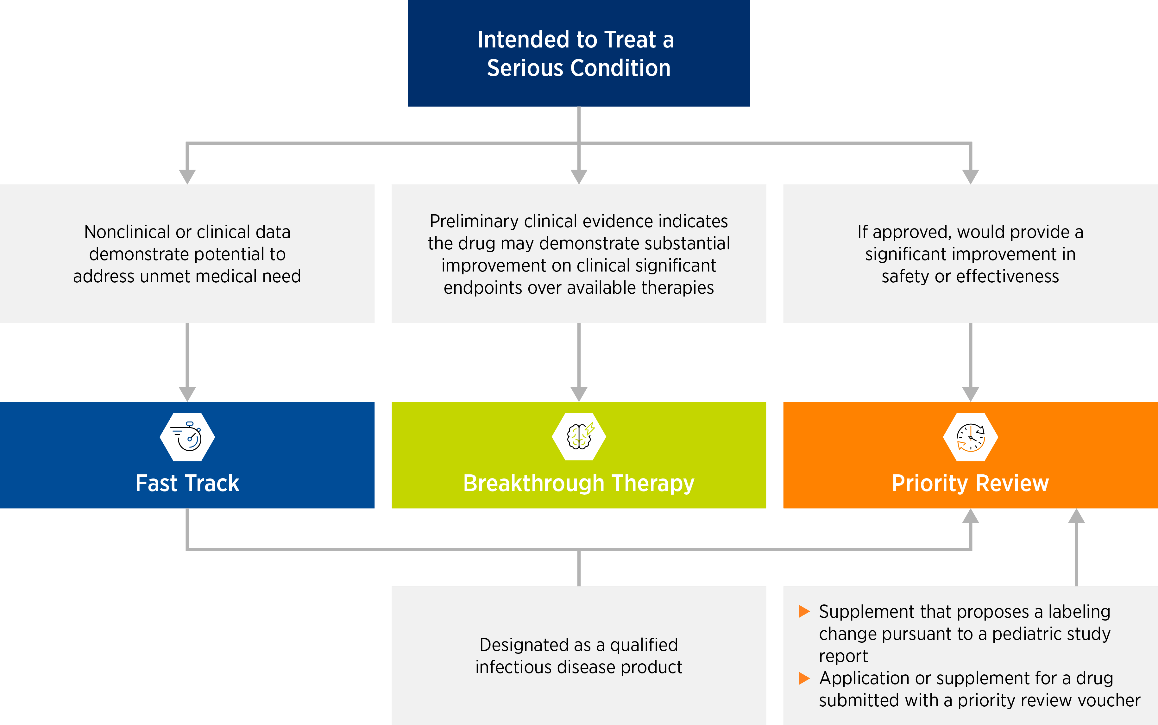
Figure 2. Qualifying criteria for designations to accelerate development
Accelerated Approval Pathway
To qualify for accelerated approval, a drug must fulfill three key criteria (see Figure 3).
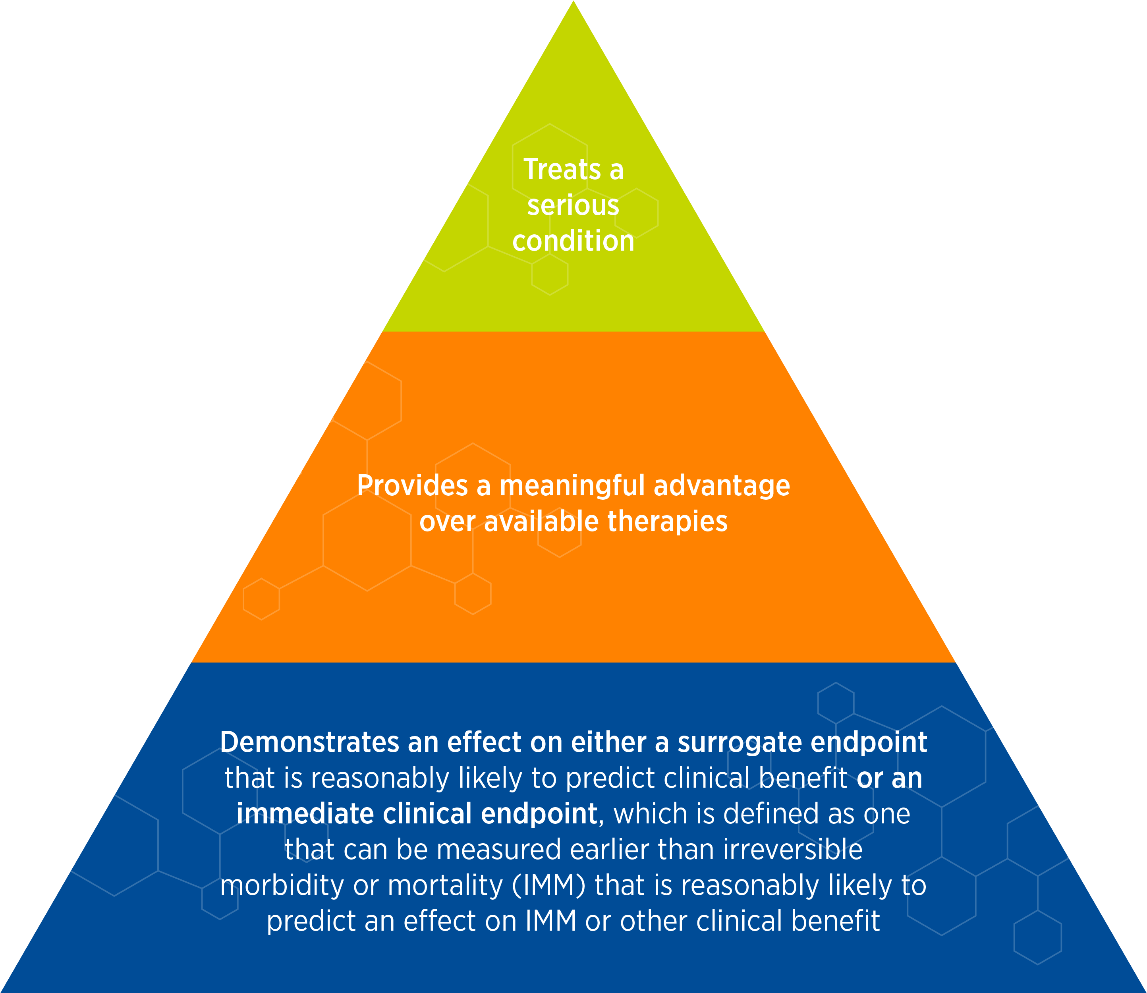
Figure 3. Qualifying criteria for accelerated approval pathway
Unlike the expedited programs for designations, there is no specified timeline for FDA response. Sponsors who are interested in pursuing accelerated approval should discuss their plans with the agency’s review division during development. Typically, they will want to know that confirmatory studies will be underway at the time of accelerated approval.
In March 2023, the FDA released draft guidance on Clinical Trial Considerations to Support Accelerated Approval of Oncology Therapeutics. The guidance points out that while single-arm trial designs and response endpoints have most commonly been used to support accelerated approval in oncology, these designs have many limitations that add uncertainty to the assessment of drug safety and/or efficacy.3 Consequently, the agency states its preference for randomized controlled trials (RCTs) to support applications for accelerated approval, whether it is one trial to verify clinical benefit or separate trials—one to support accelerated approval and another confirmatory trial to demonstrate clinical benefit.3 The FDA does, however, acknowledge that there can be circumstances—for example, where there are significant concerns about the feasibility of an RCT—where a single-arm trial is appropriate.3
Making the decision for your product
Regulatory flexibility and extensive agency guidance are available, and sponsors of rare oncology therapies should carefully consider their options for expedited programs based on their specific development plan and marketing strategy. Working with a partner like Premier Research who has extensive experience in expedited programs can help to ensure eligibility and smooth execution of the studies required to accelerate drug development for rare cancers. To schedule a conversation with one of our experts, click here.
[1] International Agency for Research on Cancer. IARC highlights rare cancers to mark Rare Disease Day 2023, February 28, 2023. Available at https://www.iarc.who.int/news-events/iarc-highlights-rare-cancers-to-mark-rare-disease-day-2023/.
[2] U.S. Food and Drug Administration. Guidance Document: Expedited Programs for Serious Conditions —Drugs and Biologics. May 2014. Available at https://www.fda.gov/media/86377/download.
[3] U.S. Food and Drug Administration. Clinical Trial Considerations to Support Accelerated Approval of Oncology Therapeutics: Draft Guidance, March 2023. Available at https://www.fda.gov/media/166431/download.

 Webinar
Webinar 


 Perspectives Blog
Perspectives Blog 