Last Updated: January 7, 2026, 4 pm UTC
Antibody-drug conjugates (ADCs) represent one of the most dynamic areas of oncology drug development. By combining monoclonal antibodies, cytotoxic payloads, and chemical linkers, these therapies are designed to selectively target tumor cells while limiting systemic exposure. Each component contributes to therapeutic potential—but also adds layers of chemistry, manufacturing, and controls (CMC) complexity.
This blog examines the critical CMC factors that define ADC development, highlighting how conjugation, payloads, linkers, manufacturing, and analytical strategies come together to ensure product quality and therapeutic success.
Conjugation Strategies and CMC Implications
The way an antibody and payload are joined has direct consequences for manufacturing consistency and clinical performance. Non-specific conjugation (see Figures 1 & 2), such as lysine- or cysteine-based approaches, often yields heterogeneous mixtures with variable drug-to-antibody ratios (DARs). High DAR species can compromise solubility and stability, leading to inconsistent pharmacological activity and a narrower therapeutic index.
Figure 1: Possible Conjugation Strategies in ADC Development

Site-specific methods—including thiol-engineered antibodies, enzymatic conjugation, and UV-based strategies—offer greater control. These approaches generate more homogeneous ADCs with tighter DAR distributions, minimizing structural disruptions and supporting more reproducible processes.
Figure 2: Possible Conjugation Strategies in ADC Development
| Lysine Conjugation | Cysteine Conjugation | Site-Specific Conjugation |
From a CMC perspective, the choice of conjugation strategy determines not only product quality but also the robustness of scale-up and long-term manufacturability.
Payload Selection and Handling
ADCs typically employ highly potent small-molecule toxins—such as DNA-damaging agents, anti-mitotic compounds, transcription inhibitors, protein toxins, or radionuclides—chosen for their ability to kill cancer cells at very low concentrations. While their diversity expands therapeutic possibilities, their potency introduces significant CMC challenges.
Payloads must be synthesized and handled in specialized facilities with strict containment protocols. Their small size also heightens the risk of immunogenicity, reinforcing the need for careful analytical characterization and control.
Linker Chemistry and Stability
The chemical linker bridges biology and chemistry in ADC development, maintaining stability in systemic circulation while enabling efficient payload release inside the target cell. Non-cleavable linkers rely on proteolytic degradation, while cleavable linkers—whether acid-labile, disulfide, or enzymatically cleavable—employ mechanisms designed to trigger release under specific intracellular conditions.
Each approach introduces distinct CMC considerations, from stability testing to release assays, and must be evaluated for both manufacturability and clinical reliability.
Manufacture of ADCs
ADC production requires both biologic and small-molecule manufacturing capabilities. Three main steps define the process: bulk production of the modified monoclonal antibody, bulk production of the small-molecule API, and the conjugation step that couples the two via an appropriate linker.
Each stage generates intermediate products with their own specifications and testing requirements before moving into final formulation and fill/finish. The integration of biologic and chemical workflows adds complexity not typically seen in other modalities.
Challenges to Manufacture ADCs
Manufacturing facilities must be versatile enough to handle both fragile biologics and highly potent small molecules—sometimes across multiple sites—adding logistical challenges. Controlling conjugation conditions to preserve activity is difficult, and batch-to-batch variability can compromise product consistency. Scale-up is particularly demanding: proteins are sensitive to environmental changes, and the linker serves as a chemistry–biology interface where unexpected reactivity can occur.
These challenges make process robustness difficult to establish and maintain, reinforcing the maxim that for ADCs, “the process is the product.” Supply chain complexity and reliance on multiple CDMO partners further compound risk (see Figure 3).
Figure 3: Supply Chain Complexity in ADC Development
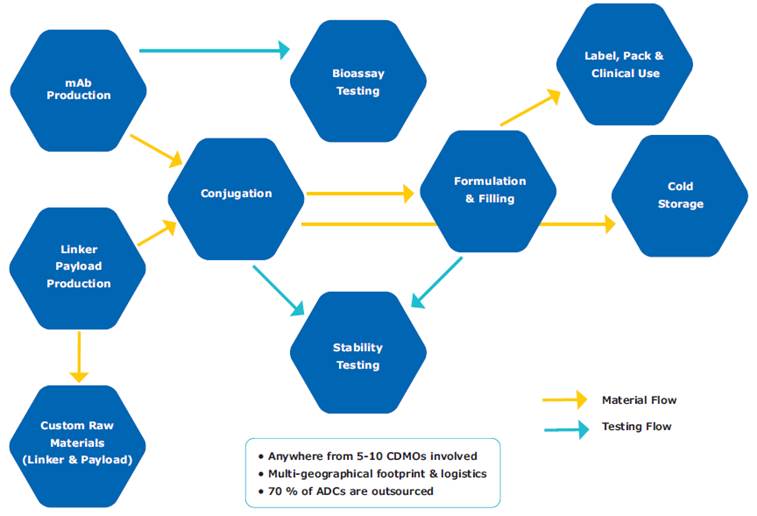
Analysis of ADCs
With three disparate components—antibody, payload, and linker—ADCs are inherently heterogeneous, demanding extensive analytical controls to ensure safety and efficacy. Characterization typically includes assessment of DAR, drug distribution, stability, pharmacokinetics, and immunogenicity.
To achieve this, multiple analytical techniques must be developed, validated, and integrated:
| Technique | Purpose |
| Liquid Chromatography-Mass Spectrometry (LC-MS) | Determines drug-to-antibody ratio (DAR), confirms molecular weight, assesses drug-loaded species |
| Hydrophobic Interaction Chromatography (HIC) | Detects hydrophobicity changes, analyzes DAR distribution |
| Reversed Phase HPLC (RP-HPLC) | Identifies impurities, evaluates DAR and drug load distribution |
| Size Exclusion Chromatography (SEC) | Assesses size distribution, detects aggregates/fragments |
| Affinity Capture LC-MS/MS | Quantifies total, conjugated, and drug-loaded antibody |
| Enzyme-Linked Immunosorbent Assay (ELISA) | Measures binding affinity to target |
| Cell-Based Bioassays | Evaluates pharmacological activity and potency |
| Ligand-Binding Assays (LBA) | Quantifies ADC analytes in biological matrices |
| Immunogenicity Assays | Detect anti-drug antibodies (ADA) |
| Stability Studies | Tests performance under ICH storage conditions |
| • pH Studies (DSC) | Evaluates thermal stability across pH environments |
| • Thermal Stability Assessment | Establishes shelf-life and degradation profile |
| • Aggregation Studies (DLS, SEC) | Monitors and quantifies aggregation tendencies |
These complementary methods provide the multidimensional controls necessary for ADC development, from early characterization through commercial release.
Future of ADCs
Next-generation ADCs are pushing boundaries in multiple dimensions: new conjugation sites and strategies to improve homogeneity, antibody fragments (fAbs) to enhance stability and reduce immunogenicity, and advanced linker technologies that better shield ADCs from immune detection. Novel payloads—including new cytotoxins, peptides, and radionuclides—are being explored, alongside dual-payload strategies, expanding therapeutic possibilities.
These innovations highlight the delicate balance between biological precision and chemical complexity. Each component—antibody, payload, and linker—introduces unique manufacturing and analytical challenges, making rigorous CMC oversight essential from early development through commercial readiness. By carefully selecting conjugation strategies, controlling DAR distributions, ensuring linker stability, and implementing comprehensive analytical programs, organizations can minimize variability, mitigate risk, and optimize therapeutic performance.
As ADC technologies evolve, the importance of robust process control, integrated manufacturing strategies, and comprehensive analytical characterization will only grow. Thoughtful planning and proactive management of these complexities are critical to translating ADC potential into consistent, safe, and effective therapies that can reach patients reliably.
Our CMC experts can help navigate the complexities of ADC development, from conjugation and manufacturing to analytical characterization. Contact our team to discuss strategies for optimizing your ADC program.
ABOUT PREMIER RESEARCH:
Premier Research, LLC (Premier) is a global leader in clinical research and consulting services with expertise in driving an efficient and effective path to market for the life sciences industry.
Premier is built with the needs of biotech in mind, turning breakthrough science into life-changing drugs, devices, and diagnostics by addressing trial complexity, overcoming development hurdles, and demonstrating product value.
Leveraging deep therapeutic expertise, innovative technology, and product development operational proficiency—from preclinical planning to clinical trial execution and commercialization—our integrated approach offers personalized, end-to-end solutions to identify the pertinent data and insight necessary to make informed decisions earlier and deliver accelerated development timelines for a smarter, faster path to approval. To learn more visit premier-research.com.
 Webinar
Webinar 


 Perspectives Blog
Perspectives Blog 