Last Updated: March 6, 2026, 9 am UTC
Infant formula is an essential food product that serves as the sole source of nutrition for many babies in North America and Europe during their first year of life, supporting health, growth, and development.[i],[ii]
As manufacturers seek to bring new products to market or support regulatory submissions for existing formulas, infant formula clinical trials have become an increasingly important—and operationally complex—component of product development.
While the U.S. infant formula shortage that began in 2022 underscored vulnerabilities in supply and regulatory readiness, the lasting impact has been a heightened focus on efficient, scalable pathways to generate high-quality clinical evidence. Sponsors planning infant formula growth monitoring studies must navigate strict enrollment requirements, early-life timelines, and caregiver burden, all while ensuring data integrity and participant safety.
Decentralized and hybrid decentralized clinical trial (DCT) models offer a compelling solution to these challenges. By reducing reliance on traditional site-based visits and shifting key activities closer to the home, DCT approaches expand geographic reach, ease participation for parents and caregivers, and support more predictable enrollment and retention. Drawing on experience from multiple infant formula studies—including fully and hybrid decentralized designs—this article outlines practical strategies for using decentralization to accelerate study start-up, improve participant experience, and support timely regulatory submissions.
In Decentralized Trials, You Can Never Be Over-Prepared
Infant formula studies face numerous challenges related to enrollment, including:
- FDA requirement to enroll infants before 14 days of life and to use formula as the sole source of nutrition for the duration of the study
- Initiatives in many hospitals are encouraging breastfeeding and disallowing the promotion of breast milk substitutes
- Reluctance of parents to bring their healthy babies to hospitals or doctors’ offices for the site visits generally required in infant formula study protocols
Since 2016, Premier has supported 8 infant formula studies, including one successfully completed hybrid decentralized clinical trial (DCT), and more than 2,000 babies enrolled. This experience includes the only infant formula GMS within FDA timelines—and the only one that did not require an extension. Based on lessons learned from our experience with decentralization approaches, we have developed tools and best practices to overcome enrollment challenges and accelerate study start-up in decentralized infant formula trials.
1. Keep the smallest of details in mind, starting with strict inclusion and exclusion criteria.
Careful consideration of inclusion and exclusion criteria helps limit screen failures. For example, for studies of cow’s milk-based infant formulas, it is useful to include prior consumption of—and ability to tolerate—such formulas in the inclusion criteria and any personal or immediate family history of cow’s milk protein allergy or intolerance in the exclusion criteria. It is also important to adhere to enrolling babies who measure within the 5th and 95th percentiles for length and weight according to the World Health Organization (WHO) growth chart.
2. Get social. Leverage the influence of social media advertising to build a pipeline of potential subjects.
For infant formula trials, it takes a lot of leads to get the number of randomized babies necessary to complete enrollment. Traditional patient recruitment firms are not effective for these studies as pregnant women may not have historically participated in clinical trials or identify with a diagnosis of pregnancy. Instead, early digital marketing efforts, consisting of targeted ads to expecting parents across the nation, are required to fill the lead funnel.
At Premier, we have tested all the major social platforms and found those that are most effective for reaching pregnant women, partners, or new parents who know they will not breastfeed. Ads on these platforms take interested individuals to a landing page with study-specific screening questions that offer them the opportunity to self-select for participation. Premier’s in-house marketing team has experience in developing marketing playbooks for Institutional Review Board (IRB) and Ethics Committee (EC) approval, allowing for a one-time approval efficiency and a mix-and-match style of advertising. This method reduces the need for additional reviews and allows the marketing team to change strategy or ad copy based on ad performance.
Based on our previous experience in recruiting for a decentralized infant formula trial in 10 metropolitan areas, enrollment rate is directly related to marketing spend. About 1,000 leads need to be generated each month to randomize 17-20 subjects, with a resulting spend of approximately $1,000 per randomized subject and an estimated 85% of randomized subjects completing the study.
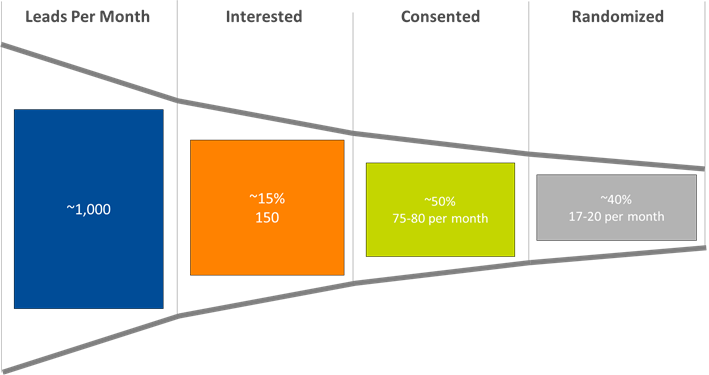
Figure 1. Subject identification funnel: from lead generation to randomization
3. Keep clarity at the forefront of study training, and reinforce through telehealth.
Anthropometrics—including measurements of weight, length, and head circumference—are integral to outcomes assessments in infant formula studies. A potential concern with decentralization is increased variability in anthropometry based on the individual performing the measurements. In our experience, the use of home healthcare nurses to perform anthropometrics did not necessarily reduce variability. Instead, training parents or caregivers to perform anthropometrics while on a telehealth visit with virtual site staff monitoring measurement performance not only increased parent/caregiver confidence but also increased consistency. Developing training videos—and making viewing of those videos part of consent—helps parents understand exactly what they are signing up for and provides them with reassurance that they can manage the responsibilities of study participation.
4. DCTs will fail without the right technologies. Deploy them simply.
Finding the right vendors for electronic patient-reported outcomes (ePROs), eDiaries, interactive response technology (IRT), electronic data capture (EDC), remote monitoring, and data management is critical to success in DCT studies. Direct data entry capability is necessary, and ePRO must be easy for parents to use. At Premier, our IRT system is pre-programmed to include the following parameters, and we recommend this approach for any infant formula study to ensure accurate randomization.
- WHO growth parameters and charts by gender and age in days to ensure accuracy at the time of enrollment.
- Expiration dates for supply of both the investigational and comparator formulas.
Remarque, our in-house study management technology platform can be used to integrate all study-related data—including administrative and financial, study management, patient, and third-party data—in one place to facilitate in-process analytics, reports, and metrics. This is particularly key for enabling the timely review of growth charts, ensuring patient safety throughout the life of the study.
5. For participants, it’s the little things that make a big difference. Continual engagement is key.
Navigating the journey from expressing interest to actually enrolling in a clinical trial takes time, so keeping in contact with parents and caregivers throughout the process is essential. At Premier, our parent support personnel routinely liaise with interested parents via email, text, or phone calls to maintain their interest in the study and to troubleshoot any issues that arise during the consent process and after randomization. With this high level of support, study teams build strong relationships with parents that continue beyond the end of the study.
Using these 5 strategies in tandem reduces enrollment timelines, increases subject retention, and accelerates study start-up, keeping trials moving forward to generate the data necessary for regulatory submissions. With an experienced CRO at the helm to oversee IRT development and maintenance, daily central monitoring, vendor management, data management, statistical analysis, and clinical study report creation, full decentralization is not only possible but preferable for all infant formula trials.
To learn more about our infant formula experience and how we can support your next program, contact us.
ABOUT PREMIER RESEARCH:
Premier Research International LLC (Premier) is a leading global clinical research organization (CRO) and consulting partner with expertise in driving an efficient and effective path to market for the life sciences industry. Built with the needs of biotech in mind, our integrated approach helps life science innovators turn breakthrough science into life-changing drugs, devices, and diagnostics by addressing trial complexity, overcoming development hurdles, and demonstrating product value.
Premier delivers a deep understanding and commitment to addressing the unique challenges of childhood diseases and pediatric development. With capabilities across the full development lifecycle, our team integrates age appropriate expertise to ensure trials are designed and conducted with children and families in mind. appropriate expertise to ensure trials are designed and conducted with children and families in mind. appropriate expertise to ensure trials are designed and conducted with children and families in mind.
Discover our pediatric clinical research experience.
REFERENCES:
[i] Helfer B, et al. Conduct and reporting of formula milk trials: a systematic review. BMJ. 2021;375:n2202.
[ii] U.S. Food and Drug Administration. FDA Developing New Framework for Continued, Expanded Access to Infant Formula Options for U.S. Parents and Caregivers, July 6, 2022.

 Webinar
Webinar 


 Perspectives Blog
Perspectives Blog 